Protein helps separate rare earths more efficiently than new tech

Penn State University scientists have discovered a new mechanism by which bacteria can select between different rare earth elements, using the ability of a bacterial protein to bind to another unit of itself, or “dimerize,” when it is bound to certain rare earths, but prefer to remain a single unit, or “monomer,” when bound to others.
By figuring out how this molecular handshake works at the atomic level, the researchers have found a way to separate these similar metals from one another quickly, efficiently, and under normal room temperature conditions. In their view, this strategy could lead to more efficient, greener mining and recycling practices for the entire tech sector.
“Biology manages to differentiate rare earths from all the other metals out there—and now, we can see how it even differentiates between the rare earths it finds useful and the ones it doesn’t,” Joseph Cotruvo Jr., lead author of the Nature paper about the discovery, said in a media statement. “We’re showing how we can adapt these approaches for rare earth recovery and separation.”
Cotruvo explained that rare earth elements, which include lanthanide metals, are in fact relatively abundant but they are dispersed.
“If you can harvest rare earths from devices that we already have, then we may not be so reliant on mining it in the first place,” Cotruvo said.
Mining and recycling
The issue is that regardless of source, the challenge of separating one rare earth from another to get a pure substance remains.
“Whether you are mining the metals from rock or from devices, you are still going to need to perform the separation. Our method, in theory, is applicable for any way in which rare earths are harvested,” he said.
Conventional rare earth separation practices require using large amounts of toxic chemicals like kerosene and phosphonates, similar to chemicals that are commonly used in insecticides, herbicides and flame retardants. The separation process requires dozens or even hundreds of steps, using these highly toxic chemicals, to achieve high-purity individual rare earth oxides.
“There is getting them out of the rock, which is one part of the problem, but one for which many solutions exist,” Cotruvo said. “But you run into a second problem once they are out because you need to separate multiple rare earths from one another. This is the biggest and most interesting challenge, discriminating between the individual rare earths because they are so alike. We’ve taken a natural protein, which we call lanmodulin or LanM, and engineered it to do just that.”
Bring in the bugs
Cotruvo and his lab turned to nature to find an alternative to the conventional solvent-based separation process, because biology has already been harvesting and harnessing the power of rare earths for millennia, especially in a class of bacteria called “methylotrophs” that often are found on plant leaves and in soil and water and play an important role in how carbon moves through the environment.
Six years ago, the lab isolated lanmodulin from one of these bacteria, and showed that it was unmatched—over 100 million times better—in its ability to bind lanthanides over common metals like calcium. Through subsequent work, they showed that it was able to purify rare earths as a group from dozens of other metals in mixtures that were too complex for traditional rare earth extraction methods. However, the protein was less effective at discriminating between the individual rare earths.
Cotruvo explained that for the new study, the team identified hundreds of other natural proteins that looked roughly like the first lanmodulin but homed in on one that was different enough—70% different—that they suspected it would have some distinct properties. This protein is found naturally in a bacterium (Hansschlegelia quercus) isolated from English oak buds.
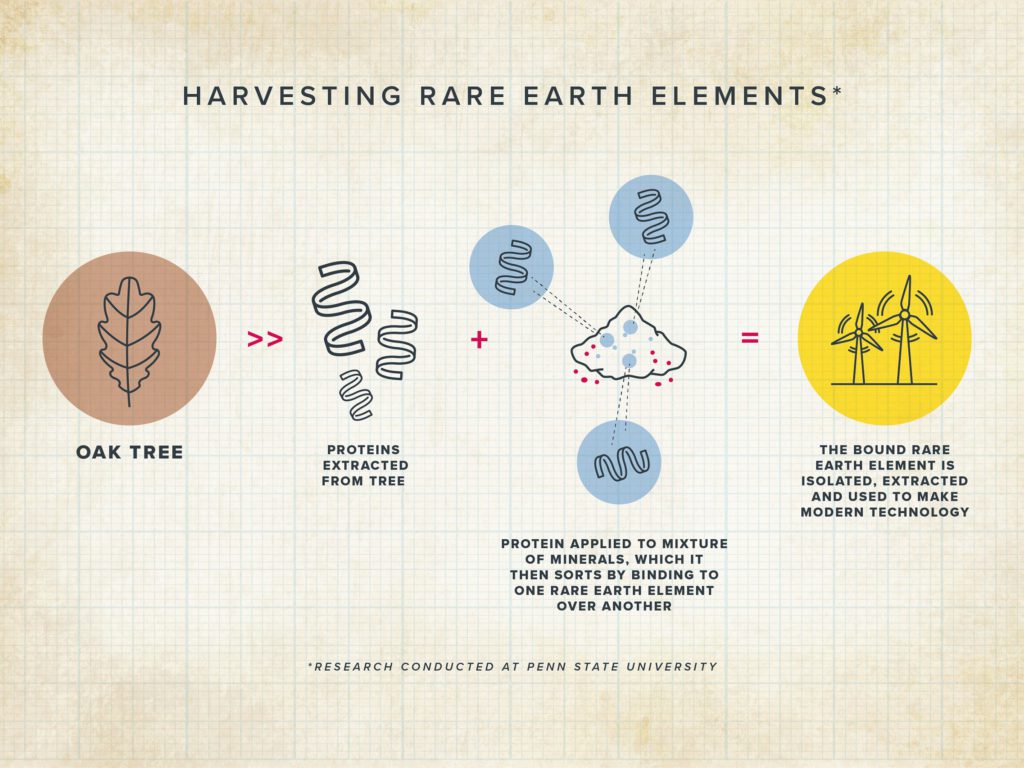
The researchers found that the lanmodulin from this bacterium exhibited strong capabilities to differentiate between rare earths. Their studies indicated that this differentiation came from the ability of the protein to dimerize and perform a kind of handshake.
When the protein binds one of the lighter lanthanides, like neodymium, the handshake (dimer) is strong. By contrast, when the protein binds to a heavier lanthanide, like dysprosium, the handshake is much weaker, such that the protein favours the monomer form.
“This was surprising because these metals are very similar in size,” Cotruvo said. “This protein has the ability to differentiate at a scale that is unimaginable to most of us—a few trillionths of a meter, a difference that is less than a tenth of the diameter of an atom.”
To visualize the process at such a small scale, the researchers used X-ray crystallography, which allows for high-resolution molecular imaging.
They determined that the protein’s ability to dimerize dependent on the lanthanide to which it was bound came down to a single amino acid—1% of the whole protein—that occupied a different position with lanthanum (which, like neodymium, is a light lanthanide) than with dysprosium.
Because this amino acid is part of a network of interconnected amino acids at the interface with the other monomer, this shift altered how the two protein units interacted. When an amino acid that is a key player in this network was removed, the protein was much less sensitive to rare earth identity and size. The findings revealed a new, natural principle for fine-tuning rare earth separations, based on the propagation of miniscule differences at the rare earth binding site to the dimer interface.
Using this knowledge, collaborators at the Lawrence Livermore National Laboratory showed that the protein could be tethered to small beads in a column and that it could separate the most important components of permanent magnets, neodymium and dysprosium, in a single step, at room temperature and without any organic solvents.
Cotruvo believes that the concept of binding rare earths at a molecular interface, such that dimerization is dependent on the exact size of the metal ion, can be a powerful approach for accomplishing challenging separations.
“This is the tip of the iceberg,” he said. “With further optimization of this phenomenon, the toughest problem of all—efficient separation of rare earths that are right next to each other on the periodic table—may be within reach.”
{{ commodity.name }}
{{ post.title }}
{{ post.date }}




Comments